5 Ways to Spot Gut-Brain Axis Imbalances Using GI-MAP

Many patients with brain fog, low mood, anxiety, poor focus, or irritability are told, “It’s stress.” While stress can certainly play a role, mental health symptoms often have a gut component.
That’s where the gut-brain connection matters. This article breaks down the key gut-brain pathways, and the main microbial signals practitioners can evaluate using GI‑MAP®. These include patterns linked to short chain fatty acids, bile acids, and inflammatory triggers like lipopolysaccharide (LPS).
What Is the Gut-Brain Axis?
The gut-brain axis is a two-way communication network between the GI tract and the brain. Signals move in both directions, meaning the brain can impact GI function while the gut can influence mood, cognition, and stress responses.
The gut is sometimes called the “second brain” because it contains the enteric nervous system (ENS)—a dense neural network lining the GI tract.1 One of the most important highways of the gut-brain connection is the vagus nerve. It carries sensory information from the gut up to the brain and also sends signals from the brain down to the gut.
Why Gut Microbes Matter for Brain Health
The gut microbiome plays a central role in the gut-brain axis because microbes produce (or trigger the body to produce) compounds that affect the nervous system. Some are directly measured in stool, while others can be estimated based on which organisms are present.
When gut microbes are out of balance—a state known as dysbiosis—the gut-brain connection can shift in a way that supports:
- Anxiety and depression
- ADHD-like symptoms
- Migraines
- Brain fog and memory issues
- Mood and behavioral changes (irritability, aggression)
Dysbiosis has also been linked in research to neurodegenerative and neurodevelopmental conditions, including Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, and autism spectrum disorder.2
In practice, the most helpful gut-brain categories to think about are:
- Neurotransmitter and hormone signaling
- Short chain fatty acids (SCFAs)
- Bile acids
- Inflammatory triggers like LPS
- Hydrogen sulfide balance
Let’s walk through what matters most clinically and how GI-MAP can help.
1) Neurotransmitter Signaling Starts in the Gut
Gut microbes can produce neurotransmitters directly or influence intestinal cells that produce them. Common examples include:
- Serotonin
- Dopamine
- Norepinephrine
- GABA
Important nuance for practitioners: Gut-produced neurotransmitters do not cross the blood-brain barrier, but they can still influence the brain through the vagus nerve, immune signaling, and precursor availability.
The Tryptophan Connection
Tryptophan is a precursor for serotonin. It must come from the diet and be properly digested and absorbed. Gut inflammation can cause increased IDO enzyme activity, which pushes tryptophan metabolism away from the serotonin pathway and toward the kynurenine pathway. This may increase production of neuroactive compounds like quinolinic acid.
What to Look for on GI-MAP
Low commensal and keystone bacteria, as some produce neurotransmitters while others stimulate the intestinal cells to make neurotransmitters
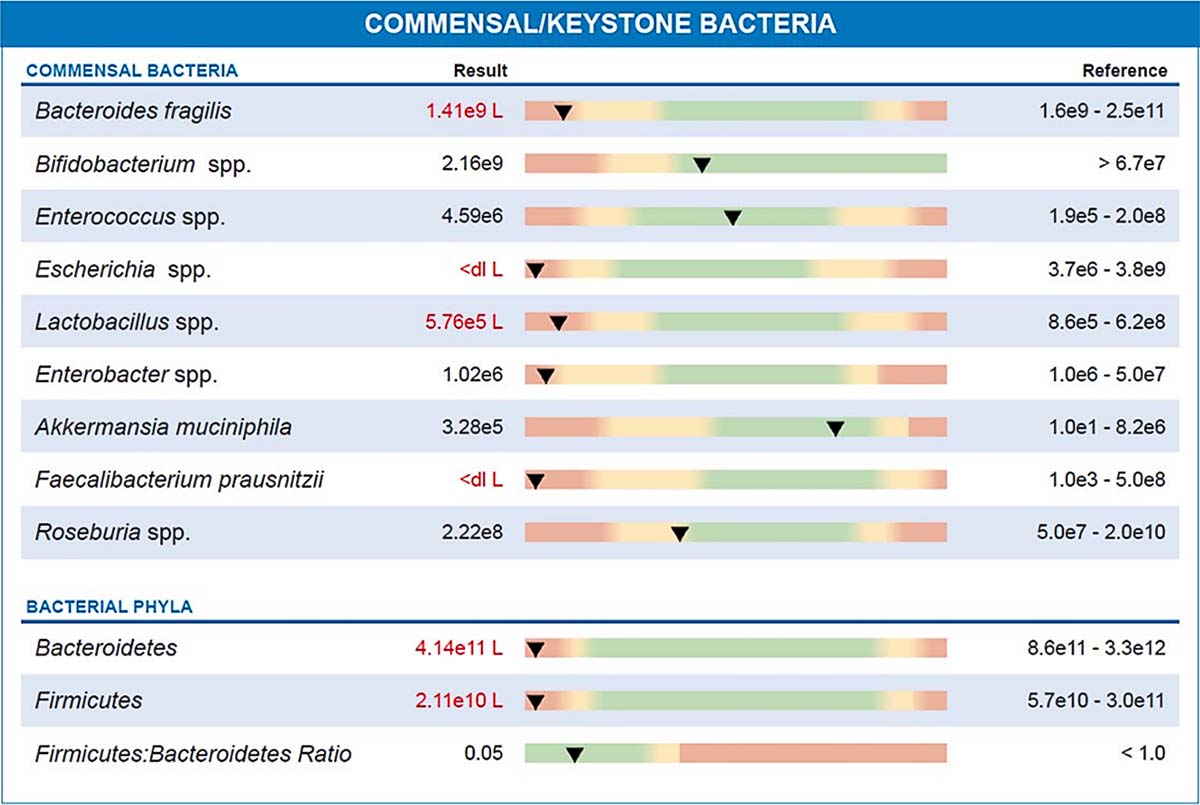
Clinical takeaway: Gut inflammation and dysbiosis can affect brain chemistry even without “classic” psychiatric triggers.
2) Short Chain Fatty Acids (SCFAs): Help Protect the Brain
Short chain fatty acids are produced when gut bacteria ferment dietary fibers and resistant starch. The main SCFAs are:
- Acetate
- Propionate
- Butyrate
SCFAs support the gut-brain connection in several ways.4-8 They can:
- Support the blood-brain barrier
- Reduce neuroinflammation
- Support neurogenesis (new neuron growth)
- Influence BDNF (a brain-supportive growth factor linked to learning and memory)
- Stimulate gut hormones like GLP-1 and PYY, which influence mood, memory, and learning
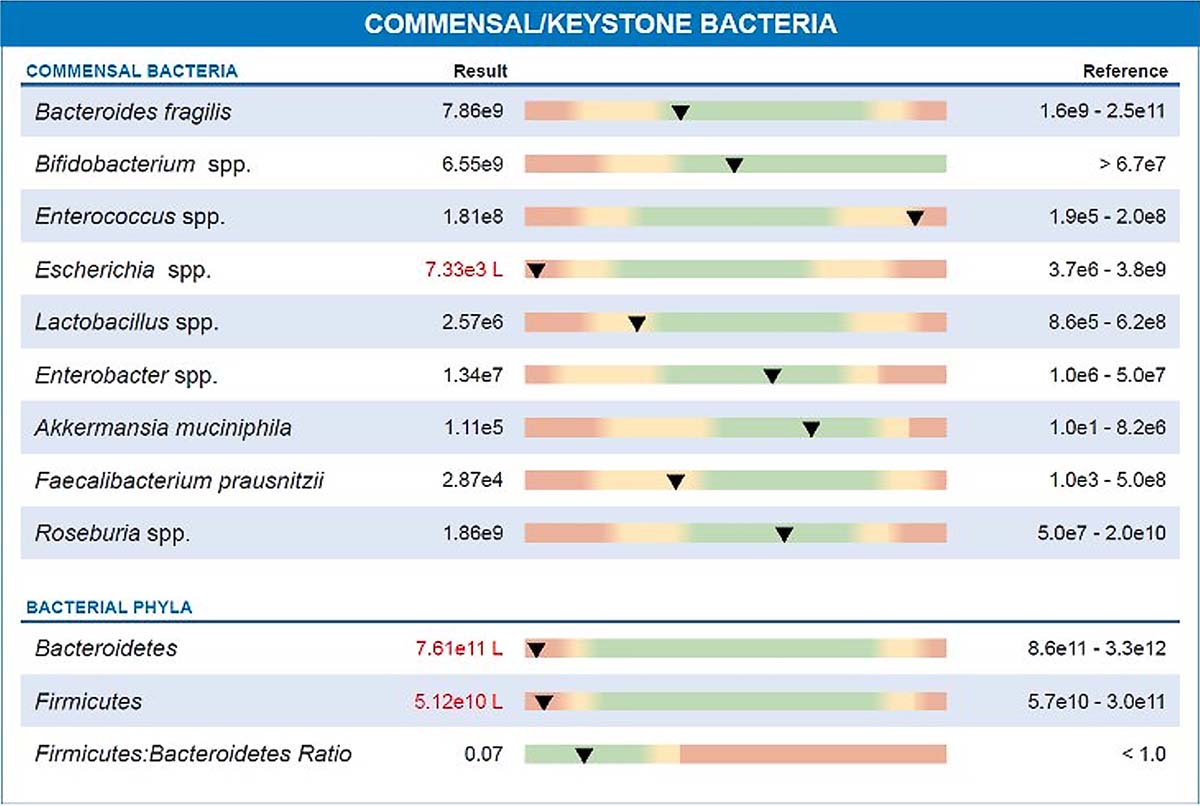
What to Look for on GI-MAP
On GI-MAP, practitioners can look for patterns suggesting low SCFA production, such as:
- Low commensals and keystone bacteria
- Low butyrate-associated markers like Faecalibacterium prausnitzii and Roseburia spp.
- Broad shifts in Firmicutes (often tied to butyrate production) and Bacteroidetes (often tied to acetate/propionate)
The StoolOMX add-on can provide direct measurement of SCFAs and their relative proportions.
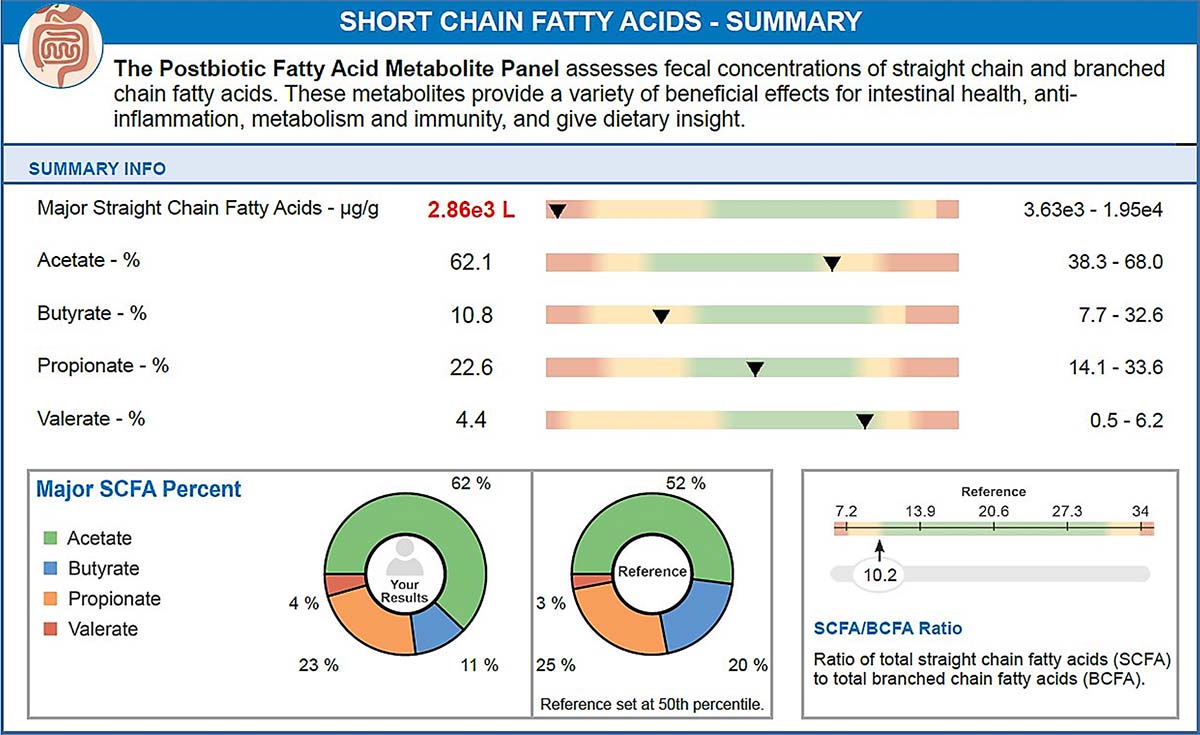
Clinical takeaway: The amount and type of fiber consumed influences the composition of the gut microbiota and consequently, short chain fatty acid production.
3) Bile Acids: A Missing Link in the Gut-Brain Axis
Bile acids are not just about fat digestion. They are also signaling molecules that can influence the nervous system.8,9
Primary bile acids are produced by the liver. In the colon, gut bacteria convert them into secondary bile acids. A small portion of bile acids can reach systemic circulation and influence the brain through signaling pathways.
Some bile acids appear more neuroprotective, while others are more irritating or cytotoxic at higher levels. Research has observed altered bile acid patterns in neurodegenerative conditions, including shifts in ratios involving secondary bile acids.10
What to Look for on GI-MAP
GI-MAP can support bile acid assessment by identifying dysbiosis patterns that may alter bile acid metabolism, including:
- Low bacteria involved in bile acid conversion (deconjugation and transformation steps)
- Dysbiosis patterns that may shift bile acid composition toward more harmful profiles

StoolOMX can provide additional insights into:
- Total bile acids
- Primary vs secondary distribution
- Conversion patterns and relative percentages
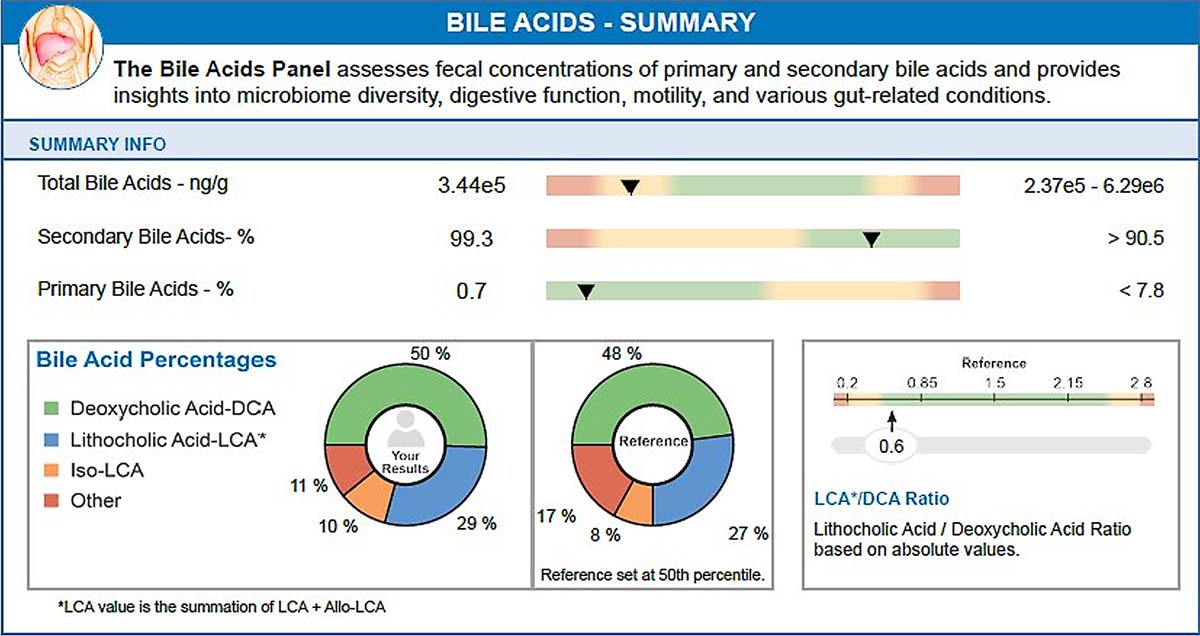
Clinical takeaway: Bile acid metabolism can be a “silent driver” of gut-brain dysfunction—especially in patients with brain symptoms plus fat intolerance, loose stools, or dysbiosis patterns.
4) LPS: When Gut Inflammation Becomes “Leaky Brain”
Lipopolysaccharide (LPS) is a component of gram-negative bacteria. In normal amounts, it’s part of gut ecology. But when gram-negative organisms overgrow, LPS exposure can rise.
Higher LPS exposure is associated with:11,12
- Increased gut permeability
- Immune activation and systemic inflammation
- Neuroinflammatory signaling
- Blood-brain barrier disruption in experimental models
What to Look for on GI-MAP
On GI-MAP, practitioners can watch for higher levels of common gram-negative and opportunistic organisms associated with LPS load, including:
- Escherichia spp.
- Enterobacter spp.
- Morganella spp.
- Pseudomonas spp.
- Citrobacter spp.
- Klebsiella spp.
- Proteus spp.
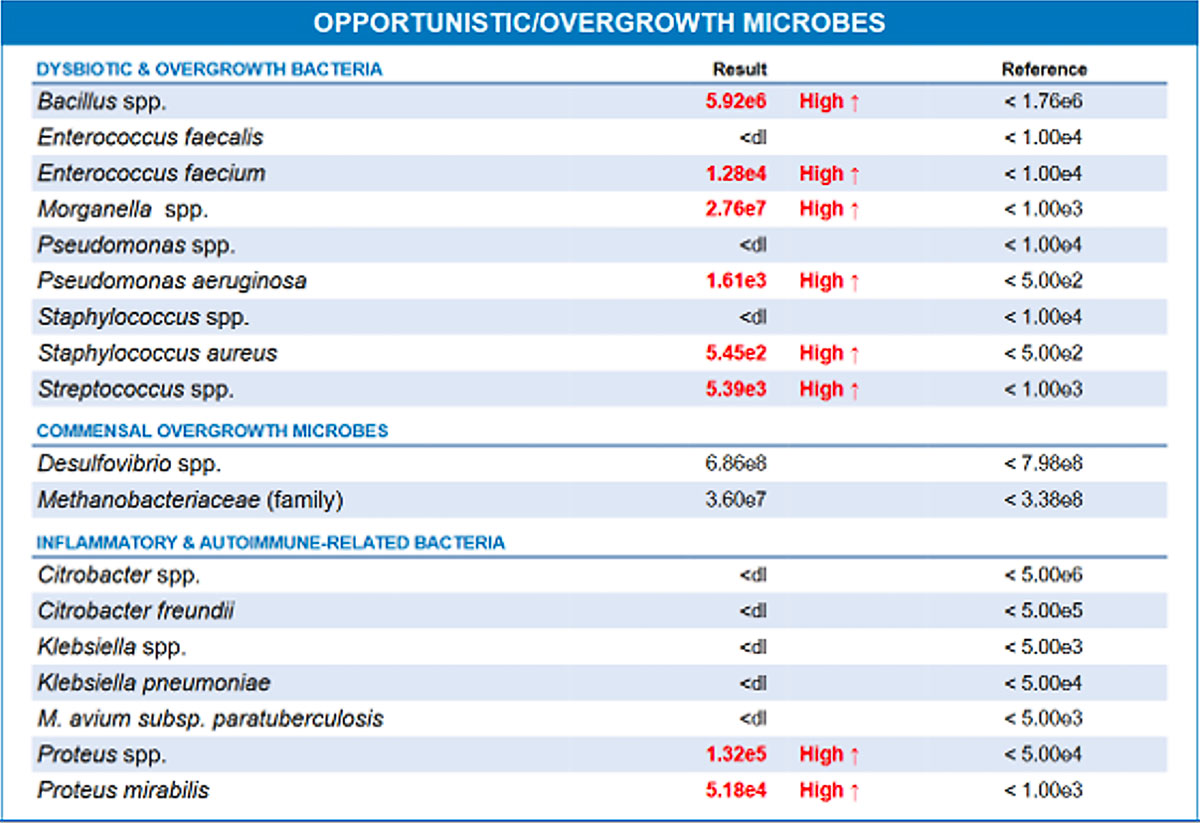
Clinical takeaway: In patients with mood issues plus inflammatory symptoms, metabolic dysfunction, or cognitive symptoms, an LPS-dominate pattern may be an important clue.
5) Hydrogen Sulfide: A “Too Much or Too Little” Signal
Hydrogen sulfide is a gut-derived gas that can influence the nervous system. Certain gut microbes can produce hydrogen sulfide via cysteine breakdown or sulfate reduction. It can be protective at normal levels and harmful at excessive levels.13-16 At normal concentrations, it acts as an anti-inflammatory, antioxidant, and neuroprotective agent. Elevated levels have been associated with memory problems, difficulty concentrating, brain fog, headaches, anxiety, and depression.
What to Look for on GI-MAP
Patterns to consider include:
- Overgrowth of hydrogen sulfide–producing organisms (Escherichia coli, Citrobacter spp., Klebsiella spp., Fusobacterium spp., etc.)
- Low or missing commensal contributors that are consistently absent (depending on context) such as Bacteroides fragilis, Escherichia spp., Desulfovibrio spp., or Fusobacterium spp.
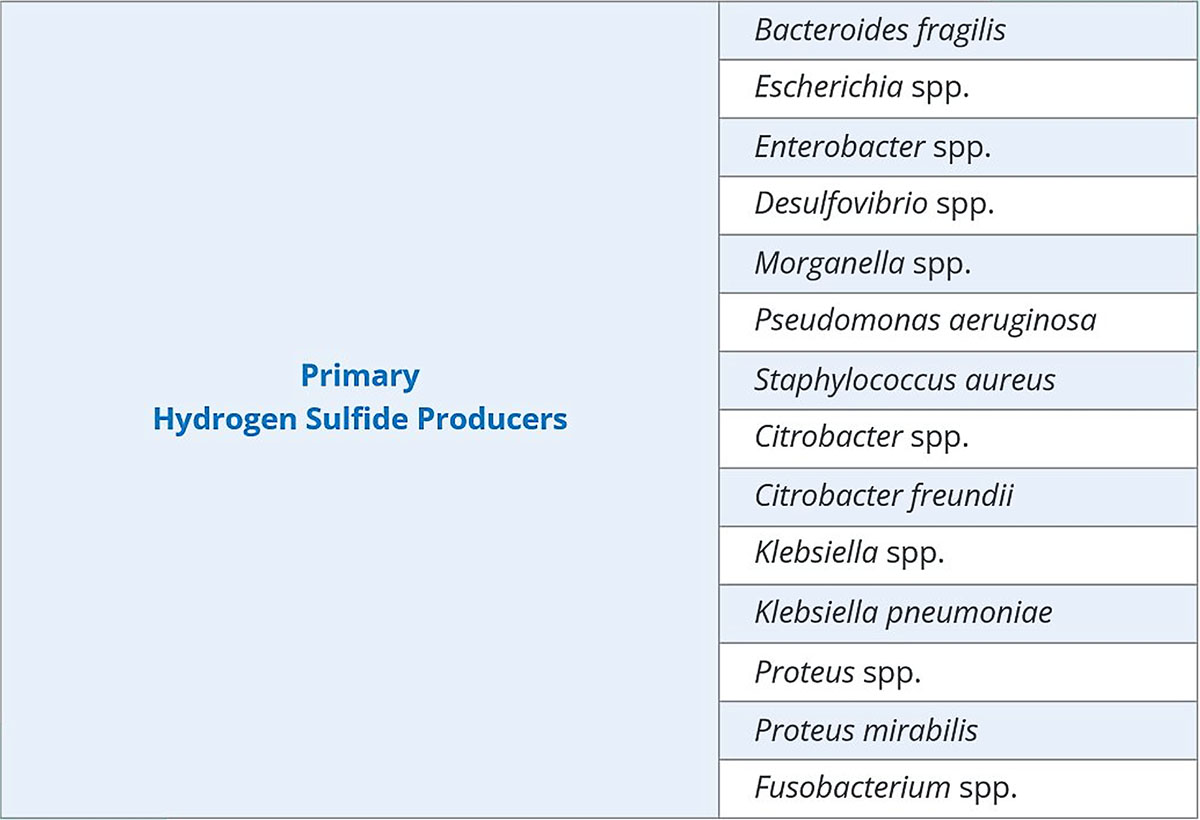
Clinical takeaway: Hydrogen sulfide is not simply “good or bad.” Patient symptoms, diet patterns, and overall dysbiosis context matter.
Pulling It Together: A Practical Gut-Brain Lens for Practitioners
The gut-brain connection rarely comes down to one organism. More often, practitioners see overlapping issues such as:
- Low commensals/keystones → reduced fermentation and signaling
- Dysbiosis → altered bile acid conversion patterns
- Gram-negative overgrowth → higher LPS signaling
- SCFA imbalance → impaired barrier integrity and neuroimmune stress
This is where GI-MAP is helpful. It lets practitioners evaluate microbial patterns tied to the gut-brain axis in one view, rather than guessing based on symptoms alone.
Conclusion
The gut-brain connection is more than a concept—it is a clinical framework. The gut-brain axis is shaped by microbes, microbial metabolites, immune signaling, and barrier integrity.
For functional medicine practitioners, GI-MAP can help identify gut patterns that may be contributing to mood, cognition, and stress-related symptoms—especially when symptoms persist and the case feels unclear.

Lisa Pomeroy, Clinical Educator
Lisa Pomeroy is a traditional naturopath with extensive training in functional medicine, lab test interpretation, and gut microbiome balancing through the Kalish Institute, The Microbiome Restoration Center, Functional Diagnostic Nutrition (FDN), Institute for Functional Medicine (IFM), The American Academy of Anti-Aging Medicine (A4M), Functional Medicine University (FMU), and others.
The opinions expressed in this presentation are the author's own. Information is provided for informational purposes only and is not meant to be a substitute for personal advice provided by a doctor or other qualified health care professional. Patients should not use the information contained herein for diagnosing a health or fitness problem or disease. Patients should always consult with a doctor or other health care professional for medical advice or information about diagnosis and treatment.
References
- Zheng Y BL, Wei T, Eleuteri AM. Understanding the Gut-Brain Axis and Its Therapeutic Implications for Neurodegenerative Disorders. Nutrients. Published 2023 Oct 31 2023;15(21):4631. doi:doi:10.3390/nu15214631
- Ullah H AS, Tian Y, et al. The gut microbiota-brain axis in neurological disorder. Front Neurosci. Published 2023 Aug 4 2023;17(1225875)doi:doi:10.3389/fnins.2023.1225875
- Mao ZH GZ, Liu DW, Liu ZS, Wu P. Gut microbiota and its metabolites - molecular mechanisms and management strategies in diabetic kidney disease. Front Immunol. Published 2023 Jan 19 2023;14(1124704)doi:doi:10.3389/fimmu.2023.1124704
- Silva YP BA, Frozza RL. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front Endocrinol (Lausanne). Published 2020 Jan 31 2020;11(25)doi:doi:10.3389/fendo.2020.00025
- den Besten G vEK, Groen AK, Venema K, Reijngoud DJ, Bakker BM. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J Lipid Res. 2013;54(9):2325–2340. doi:doi:10.1194/jlr.R036012
- Needham BD K-DR, Mazmanian SK. Gut microbial molecules in behavioural and neurodegenerative conditions. Nat Rev Neurosci. 2020;21(12):717–731. doi:doi:10.1038/s41583-020-00381-0
- Mirzaei R BB, Hosseini-Fard SR, et al. Role of microbiota-derived short-chain fatty acids in nervous system disorders. Biomed Pharmacother. 2021;139(111661)doi:doi:10.1016/j.biopha.2021.111661
- Monteiro-Cardoso VF CM, Singaraja RR. Bile Acids: A Communication Channel in the Gut-Brain Axis. Neuromolecular Med. 2021;23(1):99–117. doi:doi:10.1007/s12017-020-08625-z
- Romero-Ramírez L MJ. Emerging Roles of Bile Acids and TGR5 in the Central Nervous System: Molecular Functions and Therapeutic Implications. 2024. Published 2024 Aug 27 2024;25(17):9279. doi:doi:10.3390/ijms25179279
- Ferrell JM CJ. Bile acid receptors and signaling crosstalk in the liver, gut and brain. Liver Res. Published 2021 Jul 18 2021;5(3):105–118. doi:doi:10.1016/j.livres.2021.07.002
- Kalyan M TA, Sonali S, et al. Role of Endogenous Lipopolysaccharides in Neurological Disorders. Cells. Published 2022 Dec 14. 2022;11(24):4038. doi:doi:10.3390/cells11244038
- Brown GC HM. The endotoxin hypothesis of Alzheimer's disease. Mol Neurodegener. Published 2024 Apr 1 2024;19(1):30. doi:doi:10.1186/s13024-024-00722-y
- Zhou L WQ. Advances of H2S in Regulating Neurodegenerative Diseases by Preserving Mitochondria Function. Antioxidants (Basel). Published 2023 Mar 6 2023;12(3):652. doi:doi:10.3390/antiox12030652
- Tripathi SJ CS, Miller E, Pieper AA, Paul BD. Hydrogen sulfide signalling in neurodegenerative diseases. Br J Pharmacol. 2026;183(1):83–100. doi:doi:10.1111/bph.16170
- Munteanu C OG, Rotariu M, Poștaru M, Turnea M, Galaction AI. Role of Microbiota-Derived Hydrogen Sulfide (H2S) in Modulating the Gut-Brain Axis: Implications for Alzheimer's and Parkinson's Disease Pathogenesis. Biomedicines. Published 2024 Nov 23 2024;12(12):2670. doi:doi:10.3390/biomedicines12122670
- Kamat PK KA, Tyagi N. Role of hydrogen sulfide in brain synaptic remodeling. Methods Enzymol. 2015;(555):207–229. doi:doi:10.1016/bs.mie.2014.11.025