Autoimmune Disease: A Modern Epidemic?

There is simply no doubt that the incidence of autoimmune disorders has been rising sharply over the past several decades in the Western industrialized countries, particularly the United States (See figure 1).1 A broad array of disorders considered immune-dysregulatory and autoimmune in nature, encompassing both those classically categorized as Th1 and Th2-dominant, are included in this phenomenon. The question is why has there been such a sharp rise in the incidence of these disorders? The answers may very well be found in the current medical research, but you would probably never know it by visiting a doctor. This may be because this situation serves as an example of the giant chasm that often exists between Western medical research, which is often outstanding, and the practice of clinical medicine, which often leaves quite a bit to be desired when it comes to the management of chronic disorders with high morbidity but low mortality.
The typical allopathic clinical approach to autoimmune disorders focuses on the management of symptoms with various anti-inflammatory medications and often the use of chemotherapeutics, and very potent immunosuppressive agents with nasty potential side-effects like leukemia and lymphoma.2 While these approaches admittedly can provide substantial symptomatic relief to the patient, they do not really get to the cause of these conditions and some research suggests that these approaches may result in a furthering of the pathological process. However, modern research into autoimmune phenomenon suggest radically different approaches may be required to reverse the above cited trends, including a strong emphasis on very early detection with predictive auto-antibodies, a focus on optimizing gastrointestinal mucosal immune function and the microbiome, eradication of infectious agent triggers with antimicrobial therapy, and even the seemingly bizarre use of parasitic agents therapeutically. Some of these concepts have a long history in naturopathic and functional models of medicine, but now are emerging as hot areas of emphasis in mainstream medical research journals and investigative communities in immunology.
Molecular Mimicry
The concept of molecular mimicry is really a simple one, and it is an area attracting considerable research related to the genesis of autoimmune disorders. Simply stated, environmental exposure to specific antigens (including dietary peptides and those expressed by microbes), can in genetically susceptible individuals induce cross-reactions with structurally similar amino acid motifs associated with specific host tissues. There are now multitudes of associations that have been firmly established between immune incompatibility with specific dietary-derived antigens, as well as the overgrowth of certain opportunistic and pathogenic gastrointestinal bacteria, and the presence of specific autoimmune disorders (See table 1).3 While some of these associations have been known for quite some time, mechanisms of causality are rapidly being established in the research. However, patients suffering from disorders like rheumatoid arthritis (RA), ankylosing spondylitis (AS), and autoimmune thyroiditis (i.e., Hashimoto’s or Grave’s disease) who visit a rheumatologist or endocrinologist do not routinely have stool analysis of their GI microbiota or food sensitivity testing performed. This is ironic, particularly in the case of opportunistic microbial overgrowth in the gut, as the conventional medical paradigm typically assumes an infectious cause, doesn’t it? Perhaps this is just another example of resistance to significant change in clinical approach within medicine, even in the face of compelling evidence to do so, as it would then require a least a passive admission that something so seemingly simple was missed for so long.
Pishak et al have demonstrated that the mucous membrane of healthy people is colonized by Bifidobacteria, Lactobacilli, Bacteroides, Escherichia and Enterococci; as contrasted with the mucous membrane in RA subjects which is mainly colonized by aerobic opportunistic conventionally pathogenic enterobacteria (i.e., enteropathogenic Escherichia, Citrobacter, Enterobacter, Klebsiella, etc.), Staphylococci, Enterococci and other anaerobic bacteria (Bacteroides, Peptococci, Peptostreptococci, etc.).4 They have also reported the observed phenomenon that as RA exacerbates and then enters remission, which is a common occurrence across the spectrum of autoimmune disorders, the composition of the subject’s GI microbiota correspondingly also changes between the aberrant pattern detailed above and the one typical of normal subjects. The data of Tiwana et al suggests an increased immune response to Klebsiella in patients with AS, ulcerative colitis (UC), and Crohn’s disease (CD) and to Proteus in patients with RA.5 Alan Ebringer and his group in the United Kingdom have established over the course of many years that a substantial percentage of patients diagnosed with RA have chronic stealth infection with Proteus mirabilis in the upper urinary tract.6 His group has also established the specific amino acid motifs of cross-reaction between the Proteus hemolysin and the RA-associated HLA-DR molecules, as well as those between the Proteus urease enzyme and hyaline cartilage, containing type XI collagen, the type only found in the small joints affected in RA. His successful treatment protocol includes antibiotic therapy, such as ciprofloxacin (sometimes in combination with NSAIDs, DMARDS, and immunosuppressive agents as needed), with the added use of natural blocking agents such as cranberry juice, vitamin C for urine acidification, and plenty of fluids.7
Oral bacterial infection with Porphyromonas gingivalis, the primary cause of periodontal disease, may also play a role in peptide citrullination, theorized to be involved in the loss of self-tolerance and development of autoimmunity in RA, according to Liao et al.8 Clearly one of the challenges to the acceptance of bacterial agents as the cause of these autoimmune diseases has been that there is no universally observed association or one specific universally-causative agent. This issue is addressed head-on in research by Harkiolaki et al using a mouse model of multiple sclerosis (MS) when he states: “We show that a microbial peptide, common to several major classes of bacteria, can induce MS-like disease in humanized mice by cross-reacting with a T cell receptor (TCR) that also recognizes a peptide from myelin basic protein, a candidate MS auto-antigen. Structural analysis demonstrates this cross-reactivity is due to structural mimicry of a binding hotspot shared by self and microbial antigens, rather than to a degenerate TCR recognition. Thus, these data suggest a possible explanation for the difficulty in incriminating individual infections in the development of MS.”9 This phenomenon is also likely in play across a multitude of autoimmune disorders, and not something unique to MS.
Researchers have now gone beyond establishing mere associations between the presence of various microbes and autoimmune disorders. Some have actually experimentally induced autoimmune disease by infecting animals with specific pathogens. Mazmanian et al inoculated a wild-type mouse with the bacterium Helicobacter hepaticus to create an experimental mouse version of the autoimmune disorder inflammatory bowel disease (IBD).10 H. hepaticus activates Th17 cells which release cytokines associated with inflammation, such as IL-17, which cause symptoms of the disease. They then introduced Bacteroides fragilis expressing the polysaccharide A (PSA) to the gut of the animals where the PSA molecule was taken up by dentritic cells and presented on their surface, activating CD4 T cells and regulatory T cells (Tregs). The Tregs release IL-10 which suppresses the inflammatory action of IL-17, alleviating the IBD in mice. In summary, the researches induced autoimmune disease by introducing specific bacteria to the gut, and resolved it by introducing another, making a compelling argument for a causal relationship between the GI microbiota and autoimmune activity.
Autoimmune thyroid disorders also have been linked to bacterial infections, mainly GI overgrowth of the opportunistic organism Yersinia enterocolitica. Petru et al state; “Yersinia shows on its surface saturable binding sites for TSH. TSH receptor antibodies could be produced in selected individuals having been infected with bacteria showing TSH receptors. It may, therefore, be assumed that the gram-negative bacterium Yersinia enterocolitica may have an active part in triggering immunogenic thyroid diseases.”11 Other researchers have shown a much higher prevalence of Yesinia serum antibodies in patients with thyroid disease versus controls. However, once again, there is no universal causality established, as autoimmune phenomena is a complex issue and seems to be potentially fueled by a multitude of potential antecedents, triggers, and mediators. For example, dietary antigens have also been linked to autoimmune thyroid disease. Celiac patients have approximately 10 times the rate of auto-immune thyroid diseases (such as Hashimoto’s thyroiditis and Grave’s disease) as non-celiac individuals, reflective of the affinity of gluten-gliadin antigen-antibody complexes for thyroid tissue.12 It may be no coincidence that the emergence of an apparent epidemic of autoimmune diseases has corresponded with the ever-increasing consumption of poor-quality modern processed foods known to both negatively alter the GI microbiome and to contain a constant (often hidden) stream of offending dietary antigens, including gluten-containing grains.
While all of these associations may be interesting to researchers, what does this really mean to a clinician? Some critics would argue that there is a lack of interventional data to suggest eradication of these associated organisms and/or avoidance of these dietary antigens positively affects patient outcomes. This may be true in some instances, but it has been well established, for instance, by Ebringer that successful treatment of Proteus clinically helps those with RA,7 and dietary elimination of gluten-containing grains is entirely accepted as the most viable intervention in Celiac disease. One potential issue in play is that by the time a patient is diagnosed with autoimmune disease there is often already substantial host-tissue damage. Perhaps the horse has already left the barn? However, what if potential triggers were routinely screened for and removed by health care providers, particularly in those with a family history of autoimmune disorders? The entire course of the disorder might be favorably altered, and many of these disorders might potentially never emerge clinically. In the naturopathic and functional medicine models, there is a strong emphasis on both early detection and interventions that target the underlying pathophysiologic basis and underlying dysfunction of a disease process. Therefore, in these models the goal is to take clinical actions to reduce the potential for the disease process to progress. This also seems to intuitively make sense even in those who already have established disease; even though you may not be able to undo the damage already done, you can likely - if nothing else- slow down the train. This is particularly true since the interventions required pose little or no risk and are also relatively inexpensive; including probiotics, antimicrobial botanicals and volatile oils, mucosal-supporting nutrients and botanicals, and dietary modulation. Substantially improved molecular methods to assess the GI microbiota, utilizing PCR-DNA analysis, are also now available to clinicians at relatively low cost with rapid turn-around time.13
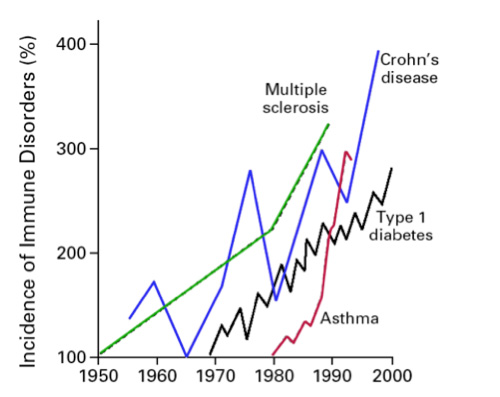
From: Bach JF. The effect of infections on susceptibility to autoimmune and allergic diseases. N Engl J Med. Sep 2002;347(12):911-920.

David M. Brady, ND, DC, CCN, DACBN, FMCP-M
Dr. David M. Brady has almost 30-years of experience as an integrative medicine practitioner and over 25 years in health sciences academia. He is a licensed naturopathic medical physician in Connecticut and Vermont, is board certified in functional medicine and clinical nutrition, a fellow of the American College of Nutrition, and completed his initial clinical training as a doctor of chiropractic...
REFERENCES
- Bach JF. The effect of infections on susceptibility to autoimmune and allergic diseases. N Engl J Med. Sep 2002;347(12):911-920.
- Inaba M, Ushijim S, Hirata N, et al. Methotrexate-related lyphomatoid granulomatosis in a patient with rheumatoid arthritis. Nihon Kokyuki Gakkai Zasshi (Article in Japanese). Aug 2011;49(8):597-601.
- Mayes MD. Epidemiologic studies of environmental agents and systemic autoimmune diseases. Environ Health Perspect 1999;107(suppl. 5):743-748
- Pishak OV. Bukovian State Medical Academy, Public Health Ministry of Ukraine. Mikrobiol Z. Sep-Oct 1999;61(5):41-47.
- Tiwana H, Wilson C, Walmsley RS, et al. Antibody responses to gut bacteria in ankylosing spondylitis, rheumatoid arthritis, Crohn’s disease and ulcerative colitis. Rheumatol Int. 1997;17:11-16.
- Ebringer A, Rahid T. Rheumatoid arthritis is an autoimmune disease triggered by Proteus urinary tract infection. Clin Dev Immunol. Mar 2006;13(1):41-48.
- Ebringer A, Rahid T, Wilson C. Rheumatoid arthritis: proposal for the use of anti-microbial therapy in early cases. Scand J Rheumatol. 2003;32:2-11.
- Liao F, Li Z, Wang Y, et al. Porphyromonas gingivalis may play an important role in the pathogenesis of periodontitis-associated rheumatoid arthritis. Med Hypotheses. Feb 2009;72;732-735.
- Harkiolaki M, Holmes SL, Svendsen P, et al. T-cell-mediated autoimmune disease due to low-affinity crossreactivity to common microbial peptides. Immunity. 20 Mar, 2009;30:348-357.
- Mazmanian SK, Round JL, Kasper DL. A microbial symbiosis factor prevents intestinal inflammatory disease. Nature. 29 May 2008;453(7195):620-625.
- Petru G, Stunzner D, Lind P, et al. Antibodies to Yersinia enterocolitica in immunogenic thyroid diseases. Acta Med Austriaca (Article in German). 1987;14(1):11-14.
- Anasaldi N, Palmas T, Corrias A, et al. Autoimmune thyroid disease and celiac disease in children. J Pediatr Gastroenterol Nutr. Jul 2003;37(1):63-66.
- Brady D. Novel Options in GI Diagnostics: DNA Detection of Gut Microbiota. Complementary Med. Jul-Aug 2008:28-31.